Program Overview
Our Certificate Course in Clinical Research Management CCRM Program is built around real-world workflows, not textbook theory. We train you on how modern trials actually run - ICH-GCP, NDCT 2019, Risk-based monitoring, CRF/EDC understanding, CDM concepts, Safety signals and Reporting, Endpoint analytics, even how AI is transforming Protocol interpretation and Data mapping.
🎯
Think Like an Industry-Ready Professional
You should think like an Industry-ready professional, not a fresher waiting for a chance. The industry wants people who understand Clinical Data.
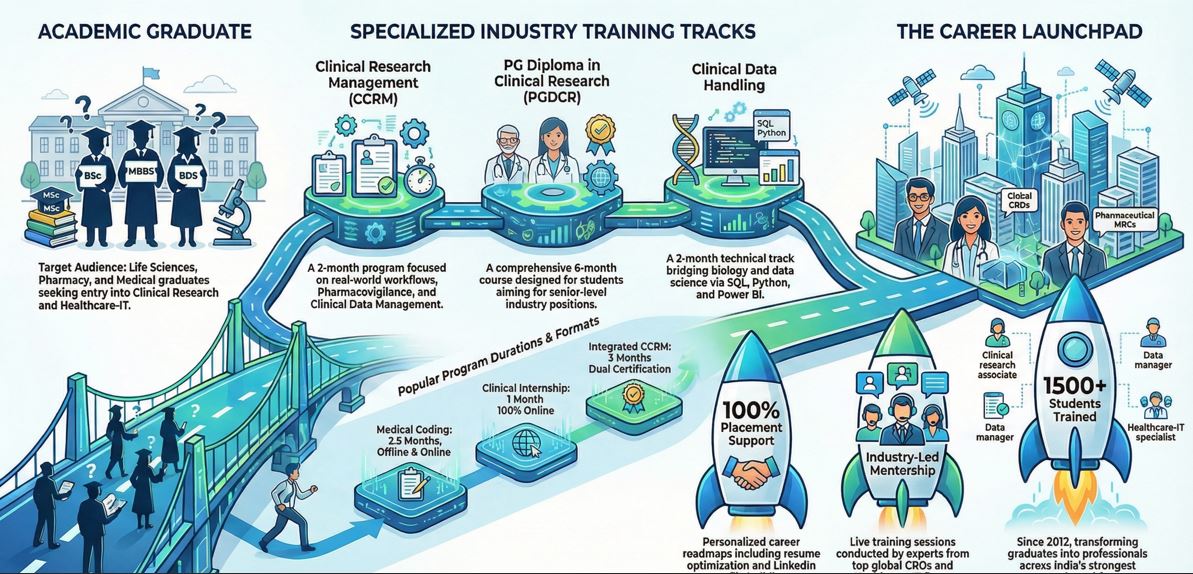
Complete Clinical Research Workflow: From Protocol to Analysis
Who Can Join?
CCRM is designed for Life Sciences, Pharmacy, Medical, and Allied Health Sciences graduates and postgraduates who want to build industry-ready clinical research careers.
💊
Pharmacy & Medical-Para Medical
🏥
Allied Health Sciences Graduates Postgraduates